By Julian Caicedo

Julián Caicedo is a Colombian engineer. For the last 20 years has been experimenting with cannabis cultivation. In 2013 he founded "Anandamide Gardens", a firm which is pioneering research into the medicinal use of cannabis in Colombia.
Julián Caicedo is associated with the BIMAC (Molecular biology, environmental and cancer group) and the GEA (Environmental studies research group) at the University of El Cauca. His work involves gathering, systematising and analysing clinical data with doctors from throughout the country who have databases of thousands of patients to monitor response to treatments with different cannabinoid ratios.
He has participated in cannabis symposia around the world and is an active member of the IACM (International Association for Cannabinoid Medicines). He was a founder member of the 'Grupo Curativa' (Group Healing) and works in collaboration with Fundación Daya in Chile and CBD Project in California. His is currently developing protocols for clinical trials and research with the Curativa group and working on educational information on the endocannabinoid system as well as running Anandamide Gardens.
In recent years, there has been increased interest in certain CBD-rich strains of cannabis for treating a wide range of human conditions in several countries. Legislators are starting to relax the restrictions on medicinal cannabis and professionals from the health field are increasingly looking at the potential of therapy using secondary metabolites of the cannabis plant.
In Colombia, the latest reforms of the regulations and laws on medicinal cannabis have been promoted by local researchers, patients, carers and parents of epilepsy patients – who in many cases had exhausted the possibilities of traditional remedies without finding any effective treatment and were desperately looking for some relief from the condition, which is so difficult to treat.
The previous legislation (Act 30/1986 of the Republic of Colombia) already allowed health workers in the country to prescribe cannabis for therapeutic use. In some indigenous communities, marijuana was used in traditional healing practices, together with coca leaves and poppies. In the 1980s, Colombia suffered a long and brutal war between the drug cartels who came to dominate the international demand for coca, poppies and marijuana, making vast fortunes at the expense of thousands of lives. Ultimately, the blame lay with prohibition; marginal substance abuse became a major public health issue and was commonly associated with crime. In those circumstances, it was unthinkable for the medical community or patients to turn to cannabis as a medicinal alternative. More recently, at the end of 2015, the Colombian Ministry for Health issued a decree regulating Act 30 of 1986 for the medicinal use of cannabis derivatives and in August 2016, Congress passed a new law regulating medicinal use of marijuana and allowing commercial cultivation, processing and exportation of derived products. The Colombian government sees the new legislation as a step in changing the country's repressive anti-drug laws and orienting them more towards public health, benefitting many patients. It has called on practitioners and researchers to conduct work in this area.
Paola Pineda Villegas is a Colombian doctor, born in Medellin, who prescribed cannabis for medical purposes for many years under the old 1986 regulation. Dr Pineda first began prescribing medicinal cannabis for the treatment of pain associated with HIV, her area of specialisation. Over time, she extended her practice the treatment of arthritis, anxiety, fibromyalgia, epilepsy, migraine, and irritable bowel syndrome.
Dr Pineda's first epilepsy patient was a little girl called Valeria Rincón, who had her first seizure at the age of three months. It lasted 17 minutes and led to her being hospitalized for status epilepticus. "She did not respond well to traditional treatment with valproico acid, Keppra, clonazepam and clobazam. All those drugs gave her constant dizziness and had severe side effects, causing hypersalivation or sialorrhea. She became withdrawn, hampering her interaction with other people" says Paola Zuluaga, Valeria's mother.
When they had exhausted the possibilities of multi-medication, the girl's doctor and mother decided to supply an oral dose of cannabis oil taken from a local sativa variety. After a few months, Valeria had gone from having daily seizures to having one every three or four days. In light of this positive response to treatment, Pineda continued to adjust the dose and the frequency of seizures fell to three months and then to five. Valeria made a remarkable recovery, her mood changed and it was even possible to reduce her conventional medication. Little Valeria is now entirely free from seizures and is getting better and better. She goes to kindergarten, laughs and sings and she is an adorable little girl.

Valeria Rincón, ‘Lele’
Dr. Pineda's has over a thousand patients across the country in her database. Of these, 30% are children with neurological disorders such as Dravet syndrome, Lennox-Gastaut syndrome, West syndrome, tuberous sclerosis (TSC), Ohtahara syndrome and idiopathic generalized epilepsy. A large proportion are currently being controlled with THC and low doses of CBD while others are responding to different ratios of THC/CBD. Some children being treated with THC even appear to get worse when the CBD rates are increased. "All my epilepsy patients have some amount of THC in their formulas" reports Dr Pineda.
Dr Pineda and her collaborators have developed several oral formulas with different ratios of the THC and CBD cannabinoids. The dosage in milligrams is measured using HPLC, following a dosage protocol they have designed on the basis of their own experience and their work monitoring patients in treatment. "This is the first clinical research with cannabinoids in Colombia. It is based on studies, patient education and cooperation and above all by a responsible attitude and a commitment to help improve people's health" says Paola Pineda.
Initially, it was not possible to measure the cannabinoids in the oral formulas precisely. Interestingly, when the cannabis extract administered to Valeria and other children with epilepsy (taken from a Colombian sativa plant, "Landrace"1) was finally quantified using HPLC, it was found to be high in THC and very low in CBD. Based largely on work in the US, treatments with a high proportion of CBD have been found to help reduce seizures, whereas high ratios of THC can even induce seizures among certain patients. However, the preliminary data from Colombia suggests that many children like Valeria respond well to THC-rich strains with low CBD.
In September 2016, Dr Pineda and her team met Professor Raphael Mechoulam of the Hebrew University of Jerusalem and told him that they were treating child patients in Colombia with formulas containing more THC than CBD. Prof Mechoulam said that the only definite data on cannabis use for epilepsy involves high doses of concentrated CBD. However, as a scientist, he said he was always willing to listen to new evidence and was surprised to hear that some epilepsy patients worsened when treated with CBD.
It is true that there is a fear of THC and its possible long-term effects, especially among child patients, "but when one sees the evidence of the therapeutic benefits in these children, one realises that there is nothing to fear, the fear of THC is based on myths and disinformation; all medication must be supplied carefully and with great responsibility, but any possible undesirable side effects of THC in children are outweighed by the visibly positive effects in reducing seizures", says Dr Pineda.
It should also be remembered that many of these children suffer very serious neurological alterations which could lead to irreparable damage in the future; all pharmacological drugs used to treat epilepsy have negative short and long-term side effects. With all pharmacological medicine, it is always necessary to weigh up the benefits of reducing the incidence of seizures against the negative effects.
Local varieties of Cannabis Sativa such as "Mango Biche" and "Punto Rojo" are biologically adapted to the climate and environmental conditions of Colombia. In the fertile volcanic areas with their varied climate zones, varieties of wild cannabis have been grown for decades and although their use for the illegal drug market has eradicated much of the original genetics, it can still be found intact in some remote sites.
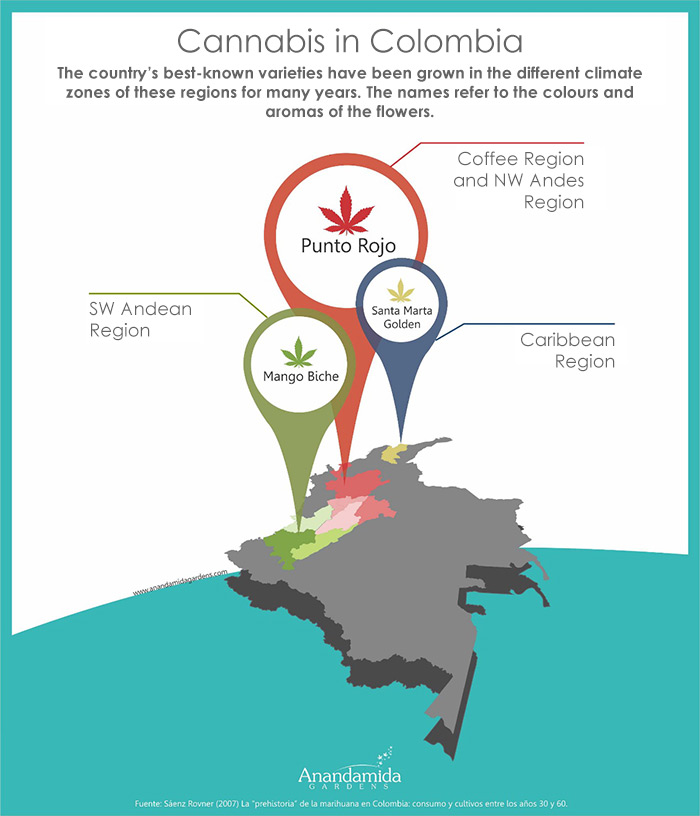
Varieties of cannabis sativa in Colombia.
Professor Raphael Mechoulam said that the natural distribution of endocannabinoids in the body might contribute to configuring the phenotypic expression of personality traits in human beings. One might also hypothesise that autochthonous varieties might be better for promoting the balance of local endocannabinoid systems, which have evolved to adapt to the local ecosystem, diet, solar irradiation, etc. This might favour more consistent interaction of the cannabis molecules with the organism in question.
THC, like CBD, could prove to be an important ally in treating neurological syndromes in children. The concept of the "Entourage Effect"2, first introduced into cannabinoid science in 1998 by Professors Raphael Mechoulam, Shimon Ben-Shabat and collaborators, suggests that the combined effect of cannabinoids may have a better therapeutic effect than the same cannabinoids in isolation. However, it is possible that the particular ratio of THC to CBD (the Cannabinoid Ratio) of local cannabis sativa varieties from a given region offer better medicinal results in the endocannabinoid systems of people from that region. As further data is compiled from clinical experiments throughout the country, more research will be needed to minimise any undesirable side effects and overcome moral objections to the use of THC in epileptic children.
It is becoming ever clearer that cannabis is a personalised medicine.
References:
1. A local variety.
2. BEN-SHABAT, Shimon, et al. An entourage effect: inactive endogenous fatty acid glycerol esters enhance 2-arachidonoyl-glycerol cannabinoid activity. European journal of pharmacology, 1998, vol. 353, no 1, p. 23-31.


