After the discovery of viruses in the latter years of the 19th century, for almost 100 years, biologists and philosophers have debated about the latter being the smallest life form known to humanity.
Relying on a strictly parasitic life-cycle, viruses can infect animals, plants, fungi and bacteria. They spread from one host cell to the other, and from one host organism to the other, and even across species; being able to morph and evolve, to increase their chance of generating a further generation.
For many years, viruses were considered the paradigm of DNA/RNA programmed for its own survival. Raw and effective, a virus "carries" and replicates a very small fragment of genetic material, as small as 3500 nucleotides (Bacteriophage MS2), necessary to encode the proteins (often enzymes) needed to subdue the host cellular metabolism, infect nearby cells, and replicate the virus, destroying the host cell.
True understanding of virus genomes and metabolism started in the '50s and progressed at great speed. Today, scientists can manipulate viruses to a fine degree: the genomes are coded on demand, and the life of the virus is managed by scientists as a puppeteer manages a puppet.
The only certainty scientists had to lose, the only minor defect in the philosophical debate from the first decades of the 20th century, is that even if viruses could be considered lifeforms, they are not the smallest known. Theodor O. Diener (US Department of Agriculture, Beltsville, Maryland, U.S) discovered the first viroid in 1971, the causal agent of a disease impacting the potato spindle tube. He showed that the agent is a free RNA of 359 nucleotides, too small to contain the genetic information necessary for self-replication, and incapable of synthesizing a shell.
VIROIDS
Viroids, the smallest known infectious agents, belong to the new order of sub-viral agents, which currently includes two families, eight genera, and 32 species of viroids. Adding another layer of complexity to the microscopic world, viruses and viroids should not be confused with one another.
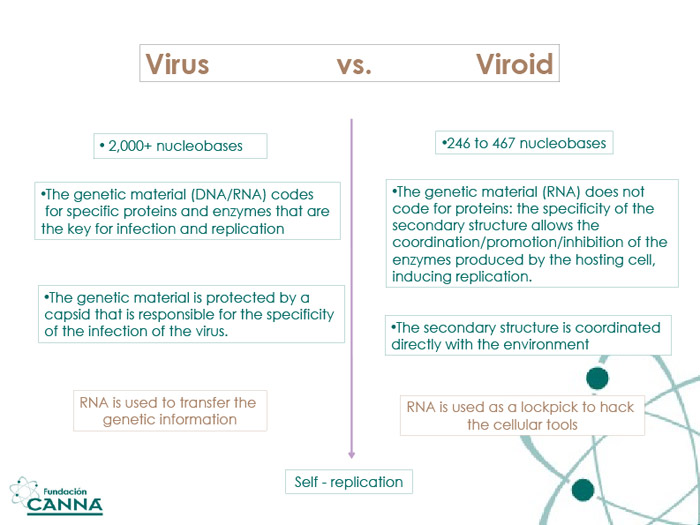
If we are willing to search for analogies, viruses can be described as "cellular hackers". After the injection of their genetic material, viruses take over the cellular machine, start synthesizing their own enzymes and proteins, and induce both self-replication and infection of nearby cells. These ancestral hackers are well protected by a capsid, a hardened shell made of proteins that is responsible for the virus specificity. Capsids give viruses their observable shape, vector viruses during their migration in organisms, and act as sensors for specific cellular membranes identified as targets. Within the capsid, the real infecting algorithm is coded as DNA/RNA, like a loaded weapon.
Viroids are different. Viroids are shapeshifters. Characterized and shaped just by their genetic materials (RNA), viroids are naked structures interacting with the environment. The nature, orientation, and strength of the interaction with nearby compounds can modify the geometries of the viroid, which can adapt to a great number of chemical environments.
With less than a 1/10 of virus total RNA-nucleobases, viroid sequences do not code for proteins or enzymes. The genetic material of a viroid is assembled to be a passepartout: able to twist, stretch, bend, and reversibly split. The key to understanding viroids' replication is accepting that the chaotic harmony that defines the cellular biology is disrupted by a single structure.
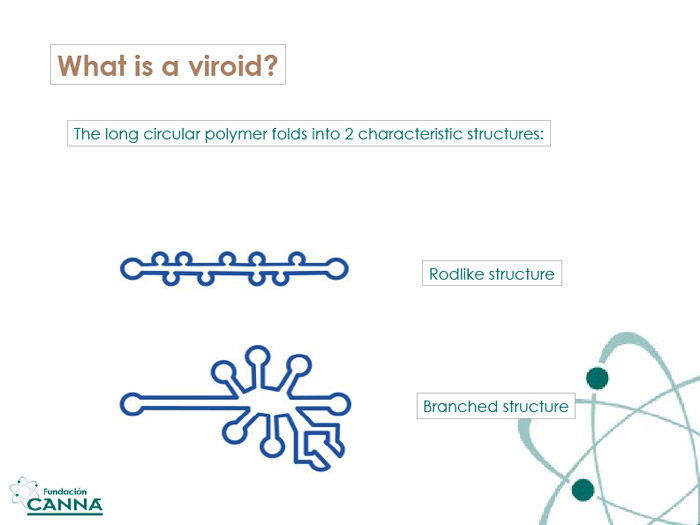
Vd-RNA is a short circular fragment of about 246-401 nucleotides, with a very high degree of base pairing. The delay between the infection of the plant and the appearance of visible symptoms characterizes the life-cycle of some viroids, which therefore lend their names to the condition.
An example of this behavior, and the focus of this chapter, is the Hop Latent Viroid, an element of the family Pospiviroidae and genus Cocadviroid.
HLVd
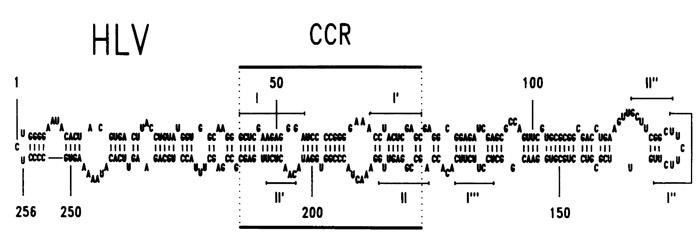
Fig.1: Original stable thermodynamic scheme of HLV, as published in Nucleic Acids research in 1988
HLVd consists of a single-stranded polymer of 256 nucleotides that assume a rod-like structure with a 65.5% degree of bases paired. As introduced in the previous chapter, HLVd is characterized by a subtle infection of hops, as the name suggests, and cannabis. Hop Latent Viroid provokes an immune response that results in necrosis, yellowing, and cell death.
The observation and characterization of HLVd in cannabis is just a few years old and is attributed to Graham Farrar of Glass House Farms with the scientific evidence produced by the University of California Davis, in collaboration with Phylos Bioscience (as a private partner). In 2017, HLVd symptoms were first noticed in California, and the viroid is now in Canada. Plants can be latent asymptomatic carriers.
HLVd symptoms
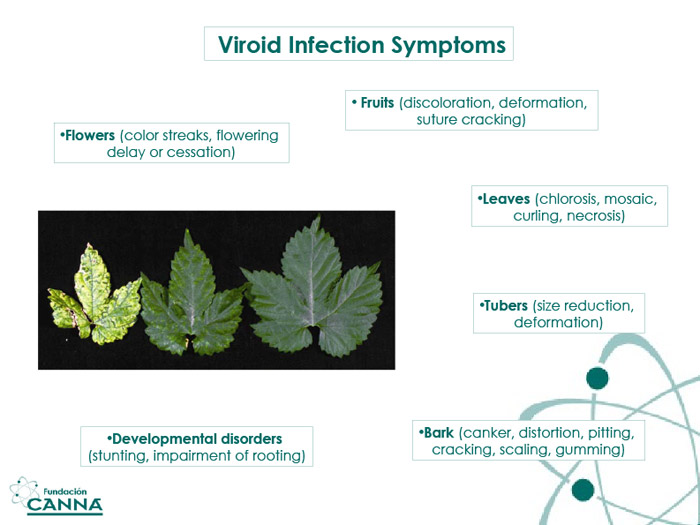
A plant with HLVd shows a set of strange symptoms: growth stunting, brittle stems, poor flowering and expression of secondary structures and metabolites, and malformation and/or chlorosis of the leaves, without an evident cause.
The latency period between the infection of the plant and the appearance of the first symptoms cannot be attributed to a single determining factor and may vary greatly between different chemo-types.
Data harvested from different studies seems to indicate that young individuals are more efficient in recognizing/fighting foreign RNA fragments as viroids, generating the delay that we rationalize as latency.
Studies around HLVd are slowly giving us an explanation for dudding, and protocols and solutions to mitigate the worldwide spread of the pathogen.
First: dudding alters the growth/defense ratio of the infected plants, meaning a generalized delay in growth times and inertia towards modifications in the growth conditions. This last effect includes reactions to nutrients, additives, and photo-period.
The second, and increasingly worrying, effect is on primary and secondary structures like flowers/fruits and hairs/spikes. For HLVd-infected cannabis plants, this means a drastic reduction in the number of trichomes and the malformation of the remaining trichomes into dry hair-like structures. The effects of the infection on modern cannabis crops are not confirmed but estimated at between a 30% and 80% loss on the yields of infected units. The number is not mitigated by the existence of a cure for living plants; HLVd infected individuals are immediately destroyed.
Knowing the dangers represented by the infectant, scientists are developing techniques to fight the viroid at cellular level and can generate a healthy offspring from an infected host with different techniques like Thermotherapy, Cold Therapy, Tissue Culture, In Vitro Micrografting, or Cryotherapy, depending on the specific viroid-host couple.
Transmission and Precautions
The recovery of clean genetic material from contaminated samples is harder than it may sound: HLVd can survive for long periods outside the comfort of living cells, and dead tissues have been proven to spread the disease.
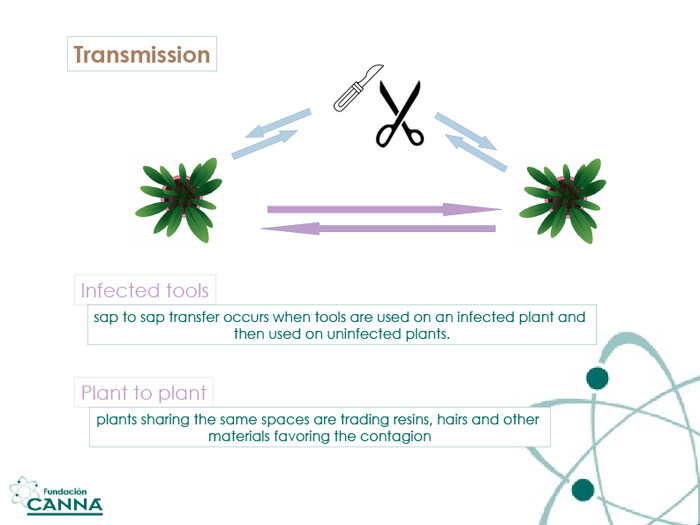
The risk of sacrificing a big fraction of the grown population is frightening enough to recommend strict hygienic protocols. The use of sterile indoor environments, mono-use gloves and tools, and the proper biological isolation of cannabis plants have been recommended by the entire scientific community since the beginning of the cannabis renaissance, but the upcoming threats will transform the recommendation into a mandatory feature. 10% bleach solution or Bunsen burner/flame sterilization are conservative ways to prevent/avoid contagion via reusable tools, limiting the expenses relative to mono-use materials.
HLVd spread in cannabis through seed is less common but cannot be excluded yet. Seeds should be considered as small "vaults" for DNA structures and are among the harshest environments for foreign genetic material, preventing the accumulation of the pathogen. The outside of the seeds could still carry debris and other fragments that may act as viroid vectors; a mindful treatment (washing) is advised to further reduce the risk.
Studies on HLVd on hops seem to exclude contagion via insects, but the data has still to be proven on cannabis variants.
Small private growers should feel relatively protected: clean indoor spaces, often populated by fewer than 10 individuals, selected from a few dozen germinated seeds or cuts, tend to minimize the risks of both plant-to-plant and tool-to-plant contagion.
On a bigger scale, the fragmentation of growing areas into smaller units and the restriction/registration of points of access in/out of growing/working spaces is GMP (Good Manufacture Practice rule) advice that should not be overlooked.
As for many other pathogens, the best way to ensure a rapid response to HLVd is a solid analytical program. Laboratories around the world are ready to identify and even sequence HLVd and other viroids/viruses with routine techniques at competitive prices.
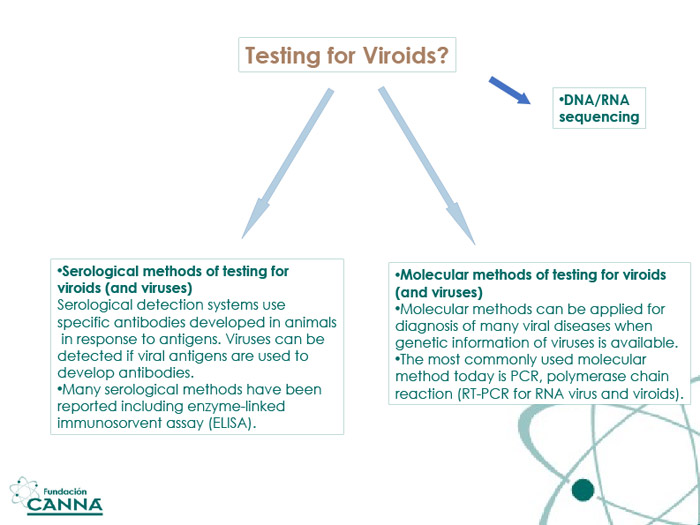
Reverse transcription PCR (RNA Polymerase Chain Reaction) is dominating the offer for commercial samples. The technique amplifies RNA sequences in the sample and, after a few passes, identifies known fragments (viroid sequences and fragments) with high precision, allowing for an early diagnosis.
RNA sequencing is also an option: the entire RNA material found in the cells is amplified, natural fragments are discarded, and the remaining ones analyzed in detail.
Prices for third party labs are decreasing rapidly, but the same techniques may be performed in-house, with specific machines.
Even with few robust methods at their disposal, scientists are developing many new different techniques to accelerate and simplify the detection of HLVd on a mass scale. In any case, multiple sample submissions may be required per plant to confidently determine that the plant is viroid-free.
To effectively diagnose a disease-free plant, some labs recommend a minimum of 3-4 tests per plant, with each test taken 1-2 weeks apart.
Growers need to be especially careful with stock plants. Generally, these samples need to be sent overnight and on ice.
If a facility tests positive for HLVd, the selected genetics may still be saved. Labs/companies have developed their own patented technique to fight viroid accumulation in specific tissues.
Heat and/or cold treatments are generally used on meristem or nodal tissue culture to clean the sample. Cleaned plant tissue cells are then grown on specialized growth media and propagated to create disease-free rooted shoots referred to as "plantlets" (also known as micropropagation).
The biggest drawback for these procedures is the price, which may reach thousands of dollars.
Conclusions
Viruses and viroids, such as HLVd, will not cease to exist just because the cannabis environment is focusing on more popular topics. Fundación CANNA is aware of the risks, connected with HLVd, that will emerge when globalization and illicit practices spread the pathogen, and it has already reacted.
Ask info@fundacion-canna.com for help!
Bibliography and Credits
Special thanks to Abha Gupta, a horticulturist, for the wonderful support in the final formulation of this article.
Gergerich, R.C., and V. V. Dolja. Introduction to Plant Viruses, the Invisible Foe. The Plant Health Instructor. 2006 DOI: 10.1094/PHI-I-2006-0414-01
Patzak, J.; Henychová, A.;Krofta, K.; Svoboda, P.; Malíˇrová, I. The Influence of Hop Latent Viroid (HLVd) Infection on Gene Expression and Secondary Metabolite Contents in Hop (Humulus lupulus L.) Glandular Trichomes. Plants 2021, 10, 2297. https://doi.org/10.3390/plants10112297
Holger Puchta, Karla Ramm and Heinz L.Sanger. The molecular structure of hop latent viroid (HLVd), a new viroid occurring worldwide in hops; Accession no. X07397 Max-Planck-Institut fuir Biochemie, April 18, 1988
Marina Barba, Zhibo Zhang. Viroid Elimination by Thermotherapy, Cold Therapy, Tissue Culture, In Vitro Micrografting, or Cryotherapy:, in Viroids and Satellites, 2017


