By José Carlos Bouso

José Carlos Bouso is a clinical psychologist and a doctor of pharmacology. His areas of interest are psychopharmacology and the therapeutic properties of entactogens, psychedelics and cannabis. He has conducted therapeutic research with MDMA, pharmacological research with several substances of plant and synthetic origin and has also performed studies on the long-term neuropsychological effects of substances such as cannabis, ayahuasca and cocaine. He is author of the book "Qué son las drogas de síntesis" [What are synthetic drugs?], and co-author of “¿La marihuana como medicamento? Los usos médicos y terapéuticos del cannabis y los cannabinoides" [Marihuana as medicine? The medical and therapeutic uses of cannabis and cannabinoids] and "Ayahuasca y salud" [Ayahuasca and health]. His research has been published in scientific journals. He is currently the director of scientific projects at Fundación ICEERS.
A total of 199 participants have begun the study in the last 2 years. To date, data have been compiled on 64 people, and we are therefore continuing to recruit volunteers.
With regard to the high number of subjects abandoning the study, it is worth stating that this most such cases (53%) occurred during the time between the first and the second assessment. As stated in the preliminary report, it is hypothesized that the reason for the high abandonment rate might be the length of the questionnaires (30-40 minutes), repeated routinely over a two-year period, which we know that some participants consider to be excessive. This would explain why the abandonment rate can be seen to stabilise following the second assessment, once participants have passed initial motivation filter and are aware of the task they will have to perform every 4 months.
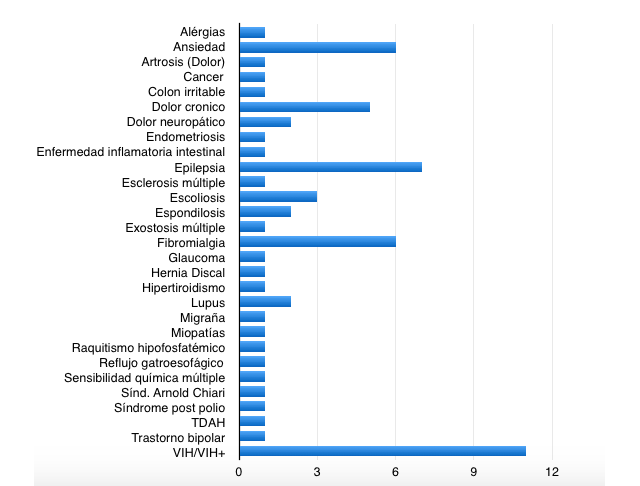
The spectrum of diseases included in our sample has changed slightly over the last 6 months; most new cases in the study consist of patients with epilepsy, bringing this condition from fourth to second place in frequency of pathologies, only behind HIV/HIV+ (See the figure below). The surveys conducted in each period are intended to gather information on aspects related to the patient’s general quality of life and other aspects specifically related to the development and evolution of the disease for which the patient is using cannabis. Making a brief and non-analytical summary of the answers received to date, it can be seen that a subjective improvement in quality of life linked to treatment with cannabis is not always determined by mechanisms of therapeutic action that directly affect the disease; rather, it is common for this improvement in quality of life to be provided by other effects, frequently attributed to cannabis, which help patients cope with the disease. Examples include, inter alia, the pro-social effects, improvement in getting to sleep, stimulation of appetite, reduction in anxiety and optimism about life. These positive effects of cannabis consumption have been drawn from the answers on positive effects in another study on psychosocial indicators of cannabis consumption which the ICEERS foundation is currently developing.
In the treatment of epilepsy, according to the interviews, cannabis offers an improvement in quality of life mainly by reducing the symptomatological expression of the pathology, i.e., by reducing the number of epileptic fits, and to a lesser extent by fostering a positive emotional state and giving a more optimistic outlook. The opposite may occur with other pathologies, where the positive effects that have the greatest impact on the quality of life are related to psychological variables and the effect that cannabis has on the symptomology of the disease in question is minimal.
We are now beginning to have longitudinal data from the first participants, who have been involved in the study since the outset 20 months ago and we will shortly be able to make the first analyses and present some preliminary results.
We are also continuing the process of recruiting new patients. If you are interested in collaborating with us, please send an e-mail to: research@iceers.org